 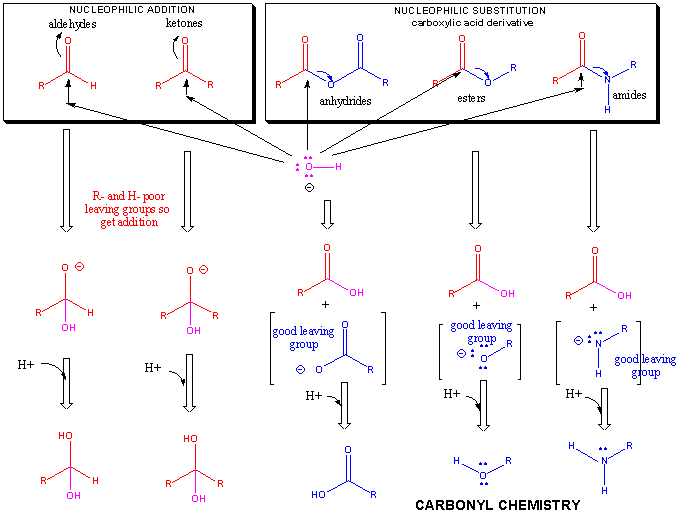
I 2 is not a nucleophile), their anions are good nucleophiles. While the halogens are not nucleophilic in their diatomic form (e.g. Similar considerations apply in the Kolbe nitrile synthesis. For this reason, the S N2 reaction of an alkyl halide with SCN − often leads to a mixture of an alkyl thiocyanate (R-SCN) and an alkyl isothiocyanate (R-NCS). For example, the thiocyanate ion (SCN −) may attack from either the sulfur or the nitrogen. If the electrophile is chiral, it typically maintains its chirality, though the S N2 product's absolute configuration is flipped as compared to that of the original electrophile.Īn ambident nucleophile is one that can attack from two or more places, resulting in two or more products. Because of this backside attack, S N2 reactions result in a inversion of the configuration of the electrophile. This means that the hydroxide ion attacks the carbon atom from the other side, exactly opposite the bromine ion. 
The bond between the carbon and the bromine then undergoes heterolytic fission, with the bromine atom taking the donated electron and becoming the bromide ion (Br −), because a S N2 reaction occurs by backside attack. In the example below, the oxygen of the hydroxide ion donates an electron pair to form a new chemical bond with the carbon at the end of the bromopropane molecule. Log 10 ( k k 0 ) = s n Types Įxamples of nucleophiles are anions such as Cl −, or a compound with a lone pair of electrons such as NH 3 ( ammonia) and PR 3. The first such attempt is found in the Swain–Scott equation derived in 1953: Nucleophiles displaying the so-called alpha effect are usually omitted in this type of treatment. The following empirical data have been obtained by measuring reaction rates for many reactions involving many nucleophiles and electrophiles. Many schemes attempting to quantify relative nucleophilic strength have been devised. Sulfur is in general a better nucleophile than oxygen. oxygen), the order of nucleophilicity will follow basicity. Within a series of nucleophiles with the same attacking element (e.g. In general, in a group across the periodic table, the more basic the ion (the higher the pK a of the conjugate acid) the more reactive it is as a nucleophile. The word nucleophile is derived from nucleus and the Greek word φιλος, philos, meaning friend. The terms nucleophile and electrophile were introduced by Christopher Kelk Ingold in 1933, replacing the terms anionoid and cationoid proposed earlier by A. Nucleophilicity is closely related to basicity. Nucleophiles may take part in nucleophilic substitution, whereby a nucleophile becomes attracted to a full or partial positive charge, and nucleophilic addition. Neutral nucleophilic reactions with solvents such as alcohols and water are named solvolysis. Nucleophilicity, sometimes referred to as nucleophile strength, refers to a substance's nucleophilic character and is often used to compare the affinity of atoms. Nucleophilic describes the affinity of a nucleophile to bond with positively charged atomic nuclei. Because nucleophiles donate electrons, they are Lewis bases. All molecules and ions with a free pair of electrons or at least one pi bond can act as nucleophiles. In chemistry, a nucleophile is a chemical species that forms bonds by donating an electron pair. A hydroxide ion acting as a nucleophile in an S N2 reaction, converting a haloalkane into an alcohol
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
